Get the facts on vaccine injuries in the U.S., like how many people are having serious reactions, filing claims and receiving compensation.
Vaccination is a life-saving medical innovation that creates no serious problems for most people. Like any other medication, vaccines can cause side effects, but the majority of immunization side effects are very mild. However, in rare situations, vaccines can cause serious injuries such as a severe allergic reaction.
This article will review vaccine injury statistics regarding these rare complications, as well as the compensation received by those who suffer immunization injuries.
Notable Statistics:
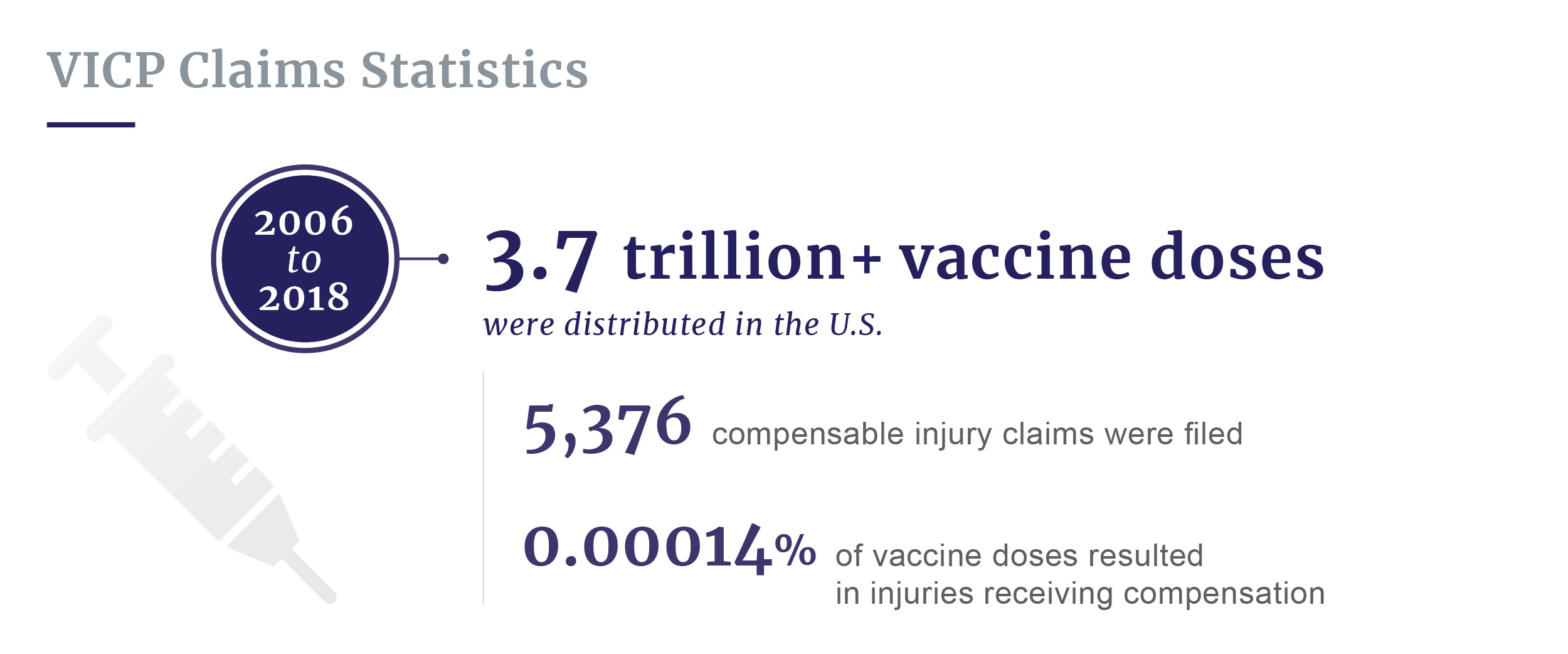
- Between 2006 and 2018, while over 3.7 trillion vaccine doses were distributed in the U.S., only 5,376 compensable injury claims were filed with the National Vaccine Injury Compensation Program (VICP).
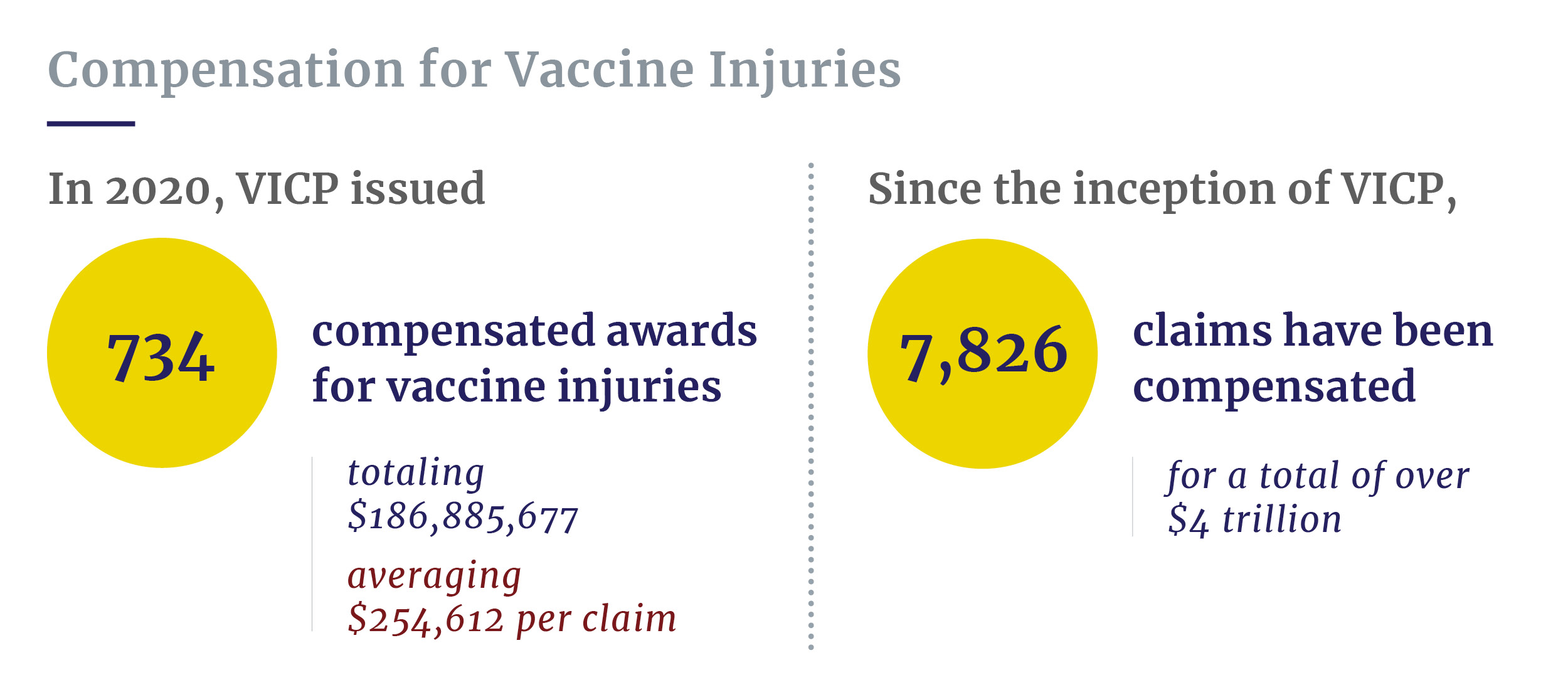
- Since the inception of VICP, 7,826 claims have been compensated for a total of over $4 trillion in awards.
- In the 2020 fiscal year, VICP paid 734 compensated awards for vaccine injuries, with award amounts totaling $186,885,678.
- In 2020, more than 46,000 reports of adverse effects following vaccinations were made to the Vaccine Adverse Event Reporting System (VAERS).
National Vaccine Injury Compensation Program
The National Vaccine Injury Compensation Program (VICP) is a no-fault alternative to traditional litigation designed to resolve vaccine injury claims. It’s administered by the Health Resources and Services Administration (HRSA), which is an agency of the U.S. Department of Health and Human Services (HHS).
The VICP was created by the National Childhood Vaccine Injury Act of 1986. The act was passed in response to the potential threats of vaccine shortages and reduction of vaccination rates caused by lawsuits against vaccine companies and health care providers.
Anyone who believes they were injured as a result of receiving a vaccine covered by VICP can file a petition. Parents can file for children, guardians can file for disabled adults and personal representatives can file for the deceased. VICP reviews the claims and provides financial compensation to people who are injured by a covered vaccine.
VICP Claims Statistics
Between 2006 and 2018, over 3.7 trillion doses of vaccines were distributed in the U.S. During this same period, only 5,376 compensable injury claims were filed with the National Vaccine Injury Compensation Program. This means about 0.00014% of vaccine doses resulted in injuries receiving compensation from VICP.
The chart below details the dispositions of VICP claims between January 1, 2006 and December 31, 2018. The “compensable claims” category includes compensation achieved through concessions by the HHS, decisions on the merits by a special master or a judge, or a settlement between the parties.
| Name of Vaccine Listed First in a Petition | Number of Doses Distributed in the U.S. | Compensable Claims | Dismissed/ Non- Compensable Claims | Total Claims |
| DT | 794,777 | 6 | 4 | 10 |
| DTaP | 105,474,077 | 159 | 126 | 285 |
| DTaP-Hep B-IPV | 74,255,807 | 42 | 57 | 99 |
| DTaP-HIB | 1,135,474 | 3 | 2 | 5 |
| DTaP-IPV | 27,884,804 | 5 | 3 | 8 |
| DTap-IPV-HIB | 68,409,736 | 16 | 39 | 55 |
| DTP | 0 | 5 | 2 | 7 |
| DTP-HIB | 0 | 3 | 1 | 4 |
| Hep A-Hep B | 16,923,878 | 18 | 7 | 25 |
| Hep B-HIB | 4,787,457 | 4 | 1 | 5 |
| Hepatitis A (Hep A) | 189,707,214 | 59 | 34 | 93 |
| Hepatitis B (Hep B) | 198,868,169 | 92 | 87 | 179 |
| HIB | 128,789,396 | 14 | 10 | 24 |
| HPV | 121,642,555 | 145 | 208 | 353 |
| Influenza | 1,672,400,000 | 3,804 | 662 | 4,466 |
| IPV | 75,537,282 | 5 | 5 | 10 |
| Measles | 135,660 | 1 | 0 | 1 |
| Meningococcal | 106,233,254 | 50 | 16 | 66 |
| MMR | 108,389,441 | 130 | 132 | 262 |
| MMR-Varicella | 28,376,497 | 24 | 16 | 40 |
| Mumps | 110,749 | 0 | 0 | 0 |
| Nonqualified | 0 | 3 | 38 | 41 |
| OPV | 0 | 1 | 5 | 6 |
| Pneumococcal Conjugate | 250,151,136 | 76 | 53 | 129 |
| Rotavirus | 116,651,363 | 46 | 17 | 63 |
| Rubella | 422,548 | 2 | 0 | 2 |
| Td | 68,120,632 | 80 | 28 | 108 |
| Tdap | 270,851,285 | 470 | 98 | 568 |
| Tetanus | 3,836,052 | 59 | 21 | 80 |
| Unspecified | 0 | 6 | 592 | 598 |
| Varicella | 121,855,108 | 48 | 22 | 70 |
| Grand Total | 3,761,744,351 | 5,376 | 2,286 | 7,662 |
In the 2020 fiscal year, VICP issued 734 compensated awards for vaccine injuries, with award amounts totaling $186,885,677.55, which averages to $254,612.64 per claim. The program also paid out an additional $20,217,550.29 in attorneys’ fees and costs payments, averaging $27,544.35 per claim.
Since the inception of VICP, 7,826 claims have been compensated for a total of over $4 trillion in awards.
Adverse Effects of Vaccination
The Centers for Disease Control and Prevention (CDC) and the Food and Drug Administration (FDA) administer a national vaccine safety surveillance program called the Vaccine Adverse Event Reporting System (VAERS).
VAERS exists to detect potential vaccine safety issues by collecting information about adverse events occurring after vaccination. Anyone, including health care professionals, vaccine manufacturers, and members of the general public can report post-vaccination health issues to VAERS.
In 2020, VAERS received more than 46,000 reports of adverse effects following vaccinations. However, fewer than 9,000 of the reports indicated that the patient scheduled a doctor’s office visit in response to the side effects, and only 4,460 reports included an emergency room or emergency department visit.
Recent Vaccine Safety Concerns
According to the CDC, despite the overwhelming benefits of vaccines, there have been a variety of historical vaccine safety concerns involving problems with specific vaccines. Most recently, in 2013, one batch of the human papillomavirus (HPV) vaccine Gardasil was recalled after a manufacturing process error. No health problems were reported.
Similarly, in 2007, Merck voluntarily recalled 1.2 million doses of Haemophilus influenzae type b (Hib) vaccines. The recall was completely precautionary and was issued due to concerns of potential contamination by the B. cereus bacteria. No evidence of B. cereus infection was identified in recipients of the recalled vaccines.
The first step in evaluating your situation is to discuss your injury with a personal injury attorney near you. Most law firms offer free initial consultations to review the facts of your case and potential legal claims you might have.